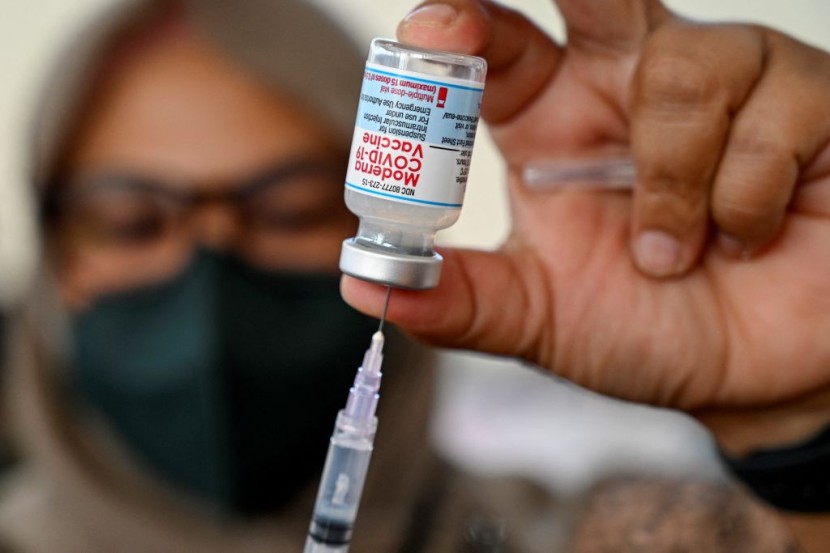
After an examination discovered stainless steel impurities in certain vials, Moderna Inc and Takeda Pharmaceutical Co Ltd said on Wednesday that they are cooperating with Japanese authorities to recall three batches of COVID-19 vaccine.
After learning of the contamination, Japanese officials halted the use of these batches of Moderna shots, which contained 1.63 million doses. Moderna said on Wednesday that stainless steel particles were found in tainted batches of its COVID-19 vaccine supplied to Japan, but the firm did not believe it constituted an undue risk to the patient's safety.
Substance discovered in Moderna COVID-19 vaccine
Hundreds of thousands of doses have been halted as a result of allegations of foreign substances discovered in vials, causing huge setbacks for the US biotech company in Japan. Authorities are also looking into the deaths of two individuals who received dosages from a contaminated batch, though the cause of death has yet to be determined, as per NDTV.
Moderna said the contamination in one of three suspended lots was traced back to manufacturing line faults at a plant managed by its Spanish contractor, ROVI Pharma Industrial Services, in a joint statement with its Japanese partner Takeda. Metallic particles of this size injected into a muscle may create a specific response, but it is unlikely to go further.
Per Reuters, the most likely source of contamination was friction between two pieces of metal in the equipment that places stoppers on the vials. It was determined that the substance was stainless steel. Moderna performed its inquiry in collaboration with Takeda and Rovi (ROVI.MC), a Spanish firm that runs the factory where the contamination happened.
Vaccination in Japan halted following the announcement
Following the announcement, Moderna's stock rose 2.6 percent. Rovi's stock was up more than 4.5 percent after dropping more than 10% on the announcement of the dosage suspensions. Takeda's stock increased by more than 2%.
In Japan, the administration of the three batches of Moderna shots was halted when 39 vials were discovered to contain foreign material. Although all of the vials originated from the same lot, doses from two other lots on the same Rovi production line were halted as a precaution.
According to Taro Kono, Japan's minister in charge of the vaccination campaign, almost 500,000 individuals have gotten injections from the three halted Moderna batches. Moderna stated that it is unable to confirm the number of doses delivered from the batches.
Both companies were alerted on August 28 that two persons had died in Japan shortly after receiving the Moderna vaccine, but there was no proof the two were linked. Local reports found no indication of the foreign material posing any health risks, and Takeda is investigating the tainted vial.
According to World Health Organization data, Japan has witnessed a fast spike in COVID-19 infections since mid-July, with 14,963 confirmed cases as of Tuesday. Moderna is investigating the tainted vials and expects to reveal its results next week. In the United States, peer-reviewed research showed the Moderna vaccination to be safe and effective in children aged 12 to 17 years old, USA Today reported.
Related Article: 15 Million COVID-19 Vaccine Doses Trashed by the United States Amid Growing Demand for Inoculations
@YouTube
© 2026 HNGN, All rights reserved. Do not reproduce without permission.








