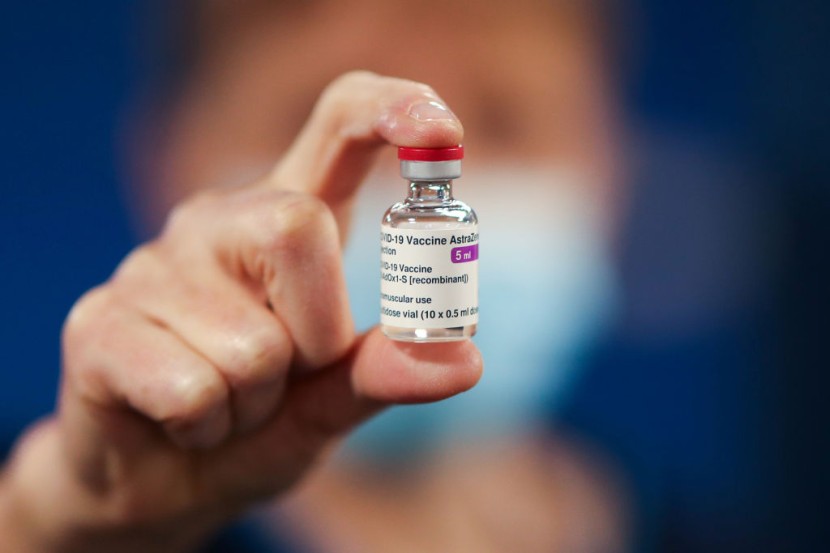
European regulators on Wednesday concluded there is a strong association between AstraZeneca's COVID-19 vaccine and life-threatening conditions involving the uncommon combination of low levels of blood platelets and blood clots. Therefore, according to the European Medicines Agency, the conditions must be listed as a "very rare side effects" of the novel coronavirus vaccine, an EU regulatory agency.
AstraZeneca Vaccine and Blood Clotting
However, it stated the benefits of being administered the vaccine shot still outweigh its risks. According to Emer Cooke, executive director of the European Medicines Agency, the vaccine's product information will include the mentioned side effects. This will be alongside a slew of other potential adverse reactions. Cooke also stated people receiving the doses should remain aware of these probable side effects, particularly within the initial two weeks of their inoculation.
A United Kingdom government advisory group stated the coronavirus vaccine should not be provided to groups of people under 30 years old. An official remarked this was out of the utmost caution because they have any serious safety concerns. Over a dozen nations suspended using the vaccine, which has been provided to tens of millions of people in Europe. This is following reports associating it to blood clots in a few dozen recipients.
Cooke remarked, notwithstanding the blood clot risk, the probing "has confirmed that the benefits of the AstraZeneca vaccine in preventing COVID-19 overall outweigh the risks of side effects." However, the EMA does not yet have the information to indicate that is true for all groups of people, according to Sabine Straus, the chair of Pharmacovigilance Risk Assessment Committee (PRAC), reported Live Science.
Fauci: Federal Government Not to Mandate COVID-19 Vaccine Passports
According to a new review by the EMA's safety committee, the vaccine remains approved in the EU for anyone 18 years above. It is the first time the regulator has confirmed a potential connection between blood clotting and the Oxford/AstraZeneca vaccine, reported Politico.
Following a study surmising 86 European cases, the EMA concluded the vaccine's benefits outweigh the risk. The report reflected information on 25 million Europeans administered with the shot.
The findings could have enormous implications for developing and middle-income nations, many of which access the AstraZeneca dose through a vaccine-sharing scheme called Covax. According to EMA officials, they looked at 18 fatalities in their analysis, reported CNN.
The majority of incidents reported within two weeks of vaccination were in women below the age of 60. With cases inundating over the previous month, numerous countries have stopped using the shot altogether.
The conclusion was based on the EMA's thorough review of 86 blood-clotting events among an estimated 25 million people inoculated with the AstraZeneca vaccine in the United Kingdom and Europe. The events reviewed by the EMA involved 62 cases of cerebral venous sinus thrombosis (CVST), an unusual form of stroke in which a clot alleviates blood from draining out of the brain.
The EMA has yet to identify specific risk factors that contributed to the phenomenon. Researchers also do not know what is specifically causing the blood clotting.
Fully Vaccinated People May Safely Resume Travel, According To CDC
© 2026 HNGN, All rights reserved. Do not reproduce without permission.








