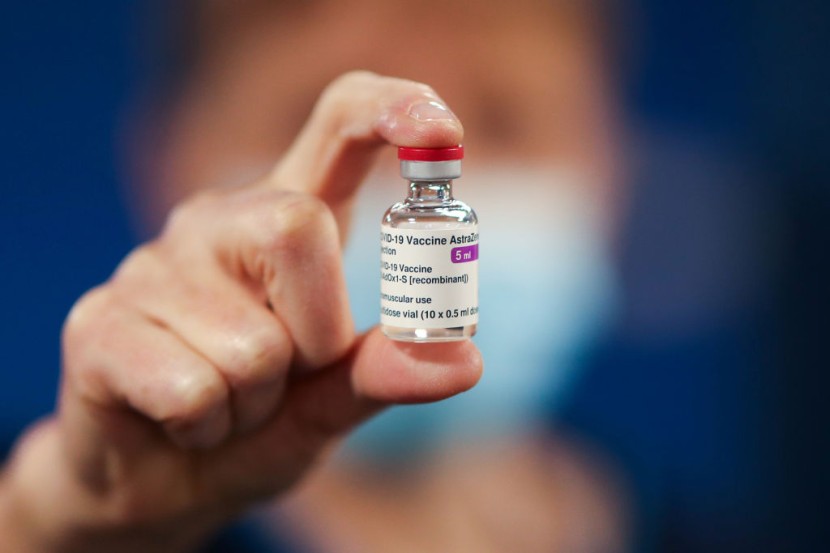
Denmark became the first nation in Europe to abandon the use of the AstraZeneca COVID-19 vaccine. This was after the country, and over a dozen other EU nations suspended its usage.
The news arrives following the discovery of experts concerning the association between the novel coronavirus vaccine and a very rare blood clotting issue. Probe into the blood clots by Denmark authorities pointed to "real and serious side effects," according to Denmark's health agency head on Wednesday.
Denmark Completely Stops Rollout of AstraZeneca Vaccine
The Danish Health Authority underscored that the doses' benefits outweigh the risks for individuals who receive them. However, they had opted to discontinue its usage due to its potential link to rare blood clotting cases and the fact that the coronavirus epidemic in Denmark is currently under wraps and other vaccines are accessible.
According to Søren Brostrøm, director-general of the Danish Health Authority, "Based on the scientific findings, our overall assessment is there is a real risk of severe side effects associated with using the COVID-19 vaccine from AstraZeneca. We have, therefore, decided to remove the vaccine from our vaccination [program]," reported NPR.
The decision was notwithstanding the World Health Organization (WHO) advisory and European medicines watchdog to continue with the vaccination. Over a dozen countries followed suit. All but a few have since resumed the usage after the European Medicines Agency (EMA) underscored the benefits of the AstraZeneca vaccine and regarded it as "safe and effective," reported Euractiv.
According to Brostrom, "Overall, we must say that the results show that there is a real and serious side effect signal in the vaccine from AstraZeneca. Based on an overall consideration, we have therefore chosen to continue the vaccination programme for all target groups without this vaccine," reported India TV.
Most Canadians Still Await Vaccination, Envy US Vaccine Roll-Out
The move will postpone the end of the Scandinavian country's inoculation scheme to early August from July 25. The WHO and EMA had consistently stated the benefits of the shot outweigh probable risks linked to its use after it was linked to numerous cases of serious blood clotting. Some of them proved fatal.
Brostrom stated the investigation reported one in 40,000 recipients of the dose would develop severe side effects. He remarked on an overall assessment that they have chosen to continue the inoculation program for all target groups without the COVID-19 vaccine.
Brostrom noted it was a "difficult decision" to make. He said the upcoming target groups for inoculation are less likely to become seriously ill from COVID-19. He added officials should weigh this against the fact they now have a known risk of serious adverse effects from inoculation with AstraZeneca, even if the risk in absolute terms is not extreme.
With the sheer number of readily accessible shots of the vaccine and limited proof of why the blood clotting might be forming, numerous nations in the European Nations and elsewhere had resumed use of the vaccine. This was because the susceptibility of dying from COVID-19 was "much greater" than the susceptibility of mortality from its rare side effects.
Japanese Woman, First COVID-19 Patient to Receive Living Donor Lung Transplant in the World
© 2026 HNGN, All rights reserved. Do not reproduce without permission.








